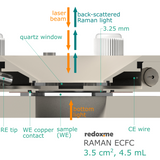
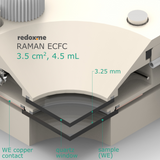
Raman ECFC 3.5 cm2, 4.5 mL – Raman Electrochemical Flow Cell, active area: 3.5 cm2, volume: 4.5 mL

This cell combines two classical analytical techniques such as electrochemistry and Raman spectroscopy, to obtain in-situ chemical information about the reactions taking place during an electrochemical experiment. It consists mainly of three elements: (i) sample holder with Tantalum (formerly Copper) foil serving as an electric contact to working electrode surface, (ii) chamber in which reference and counter electrodes are installed together with electrolyte inlet and outlet and (iii) lid which seals the cell and holds the Sapphire window. The sample consisting of a thin film of electrochemically active material deposited on a rigid or flexible substrate (working electrode) is loaded from the bottom via magnetic or screw mount. The counter or/and reference electrodes are mounted in a top casing (either 2-, or 3-electrode setup. The oval counter electrode made of e.g. Platinum wire assures uniform distribution of the field lines along the path to working electrode. During an experiment Raman laser is focused onto the surface of a thin film working electrode through a transparent Sapphire window and thin layer of electrolyte (total optical path of 3.25 mm). The electrolyte thickness of 2.25 mm ensures free diffusion of ions (e.g. protons) and its counter ions.
The electrode adapter for installing 6 mm dia. electrodes inside Raman ECFC is available here. It enables using disk electrode, plug with clip or any other rod-shape electrode (e.g. graphite rod) or current collector (e.g. metal mesh, graphite coated metal mesh, metal foil, graphite coated metal foil, metal foam, carbon woven and non-woven fabrics, carbon paper, etc.) as working electrode instead of a flat electrode in default Raman ECFC configuration.
The screw mount variant of Raman ECFC can be converted into GDE Raman ECFC by using Gas Compartment. This conversion allows installation of Gas Diffusion Electrodes as working electrode.
The cell elements are constructed with materials that are inert to the sample (PEEK, Fluorocarbons). It well fits aqueous (FKM O-Rings) and organic solvent (FFKM O-Rings) electrolyte requirements. The construction is gas-tight and can be used when the removal and exclusion of contaminants such as oxygen and water is required by bubbling of an inert gas through the electrolyte (in an external reservoir).
Application note
This cell can be used to track kinetic phenomena such as the near-surface proton concentration changes during oxidation and reduction reaction at working electrode. It can be also used to identify materials such as carbon, metal oxides, polymers and electrolytes, and to determine their structure and distribution. Various metals are suitable for this cell as auxiliary electrode including Platinum, Gold and Silver. For troubleshooting instruction see the redox.me blog post
Operating Video Manual:
Specification
nominal exposure area: 3.5 cm2
electrolyte volume: 4.5 mL
optical path (including Sapphire window): 3.25 mm
electrode plug diameter: 6 mm
Intrastat data
HS Code: 90275000
Country of Origin: Sweden
NET weight: 200g
Product includes
1 x chamber
1 x Ag/AgCl or non-aqueous Ag/Ag+ 30 mm reference electrode
1 x Metal wire auxiliary electrode – ST 0.6/150 mm, Platinum
1 x Sapphire window - 0054PSPCM
1 x lid
1 x WE Tantalum contact
1 x sample holder
1 x plug
Related products
BEC 50 mL - Basic Electrochemical Cell
Gas Compartment
Peristaltic pump