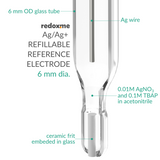
Non-aqueous Silver / Silver Ion Refillable Reference Electrode - 6 mm dia.

It is a compact and refillable non-aqueous Ag/Ag+ reference electrode equipped with ceramic frit molten into the glass body. Ag/Ag+ reference electrode consists of a silver wire immersed into an electrolyte containing silver ions. This electrode outperforms competing products in terms of mechanical stability, voltage stability, and lifetime. In general, the reference electrode is composed of a system of phases, which retain an essentially constant composition and therefore provides a stable potential by which the working electrode potential can be monitored. The phases present in reference electrodes undergo reversible redox processes at a very high rate, enabling them to rapidly adjust to changes in the solution’s ionic activity, but leaving them sensitive to the passage of large current densities. The included FFKM O-Ring well fits organic electrolyte requirements.
Application note
This type of reference electrode is commonly employed in non-aqueous electrochemical studies, i.e. whenever a solvent based electrolyte is used. The reference electrode should be stored with the rubber plug applied on the tip. Storing in the solution may shorten its lifetime (the electrode operates well as long as there is at least ¼ of the solution in the electrode tube). Before use, rinse it with acetonitrile, acetone, isopropanol, or ethanol. If air bubbles are present around the porous frit, flick the tip several times to remove them. For cleaning, use acetonitrile, acetone, isopropanol, or ethanol. To re-fill the electrode please follow this article.
Specification
effective length: 30 mm or 70 mm
tube diameter: 6 mm
filling solution: 0.01M AgNO3 and 0.1M TBAP in acetonitrile
potential stability: <5 mV
frit resistance: 2-10 kohm at RT
operating temperature: 0-75 deg C
Intrastat data
HS Code: 90278930
Country of Origin: Sweden
NET weight: 100g
Product includes
1x non-aqueous Ag/Ag+ reference electrode
1x O-ring
1x glass tube for storing purposes
Related products
Electrochemical cells
Photo-electrochemical cells