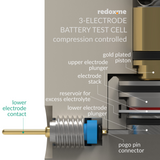
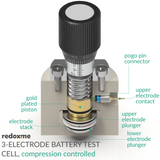
Three Electrode Battery Test Cell – compression controlled

This cell is designed as high-quality alternative to Swagelok-type constructions for reproducible electrochemical measurements of battery and supercapacitor materials. The cell consists of two main elements: (i) the cell base with micrometre screw and compression spring, which allows precise control of pressure applied to the electrode sandwich; and (ii) electrode cartridge where the anode, cathode and reference electrode are located. The (default) electrode cartridge and plungers are designed for planar electrodes with diameter of 18 mm and total sandwich thickness of 2.5 mm. The diameter of separator shall be at least 2 mm larger than electrode diameter (max. 22 mm). The lock ring secures the separator in the electrode cartridge allowing easy and safe electrodes insertion, while ensuring accurate alignment. The upper and lower electrode plungers are available in various materials: 316L Stainless Steel (default), copper, aluminum, nickel etc. The pin-type reference electrode is installed from the side of the electrode cartridge. Thanks to sharp edged hole at the end of the pin the reference electrode (Li/Na/K etc.) can be easily punched from the metal foil and directly loaded into cell. The magnetic mount facilitates rapid assembly of the cartridge in the cell base. The force applied to the electrodes can reach up to 90N and is adjusted with the micrometer control knob. For 2-electrode measurements the reference electrode feedthrough hole can be closed with the reference electrode punch.
The cell elements are constructed with materials that are inert to the sample (Stainless steel and PEEK). It well fits aqueous (FKM and EPDM O-Rings) and organic solvent (FFKM O-Rings) electrolyte requirements. Good electrical contact is ensured by gold plated pins. The construction is gas-tight and can be effortlessly assembled in the glove box, reducing possible human error to minimum.
Application note
This cell can be used for all common battery measurements, such as galvanostatic cycling, cyclic voltammetry or electrochemical impedance spectroscopy. Both liquid and solid-state or gel-polymer electrolytes can be studied. Additionally, properties and performance of supercapacitor materials can be investigated. Various materials can be examined, including typical Li-ion electrodes (graphite, NMC, LTO etc.) and other chemistries – sodium, magnesium, potassium etc. It can also be applied to measure the ionic conductivity of separators and electrolytes. Owing to the micrometer control knob, the relationship between the initial pressure applied to the cell and the performance of solid-state electrolytes or metal plating process (anode-free systems) can be studied.

The above graph shows relation of pressure applied to electrode stack and distance travelled from the point of contact. The point of contact can be determined by checking OCV (see manual video) or by calculation. To calculate the point of contact, subtract electrode stack thickness from 4 mm. For example, for the 0.9 mm stack, the point of contact (i.e. reading on micrometer screw) will be 4 mm – 0.9 mm = 3.1 mm. Therefore, you should set micrometer screw to 3.1 mm and measure distance/pressure relation from this point.
Operating Video Manual
Specification
recommended electrode diameter: 18 mm (other options: 12 mm, 15 mm, 16 mm)
recommended minimum separator diameter: electrode diameter + 2 mm (max. 22 mm)
maximum electrode sandwich thickness: 2.5 mm
spring rate: 10.86 N/mm
maximum spring load: 90 N
operating temperature: -40°C – 80°C (default)
Intrastat data
HS Code: 90309000
Country of Origin: Sweden
NET weight: 400g
Product includes
1 x cell base
1 x compression control unit
1 x Three-Electrode Cartridge
1 x reference electrode punch
1 x upper plunger (Stainless Steel)
1 x lower plunger (Stainless Steel)
1 x separator locking ring
1 x gold plated piston
1 x compression spring
2 x gold plated pin contact
1 x set of O-rings
1 x set of 3 cable adapters 1mm to 4mm banana plug
1 x electrode plunger removal tool
Related products
Battery Cartridge – three-electrode setup
Battery Cartridge – temperature monitoring